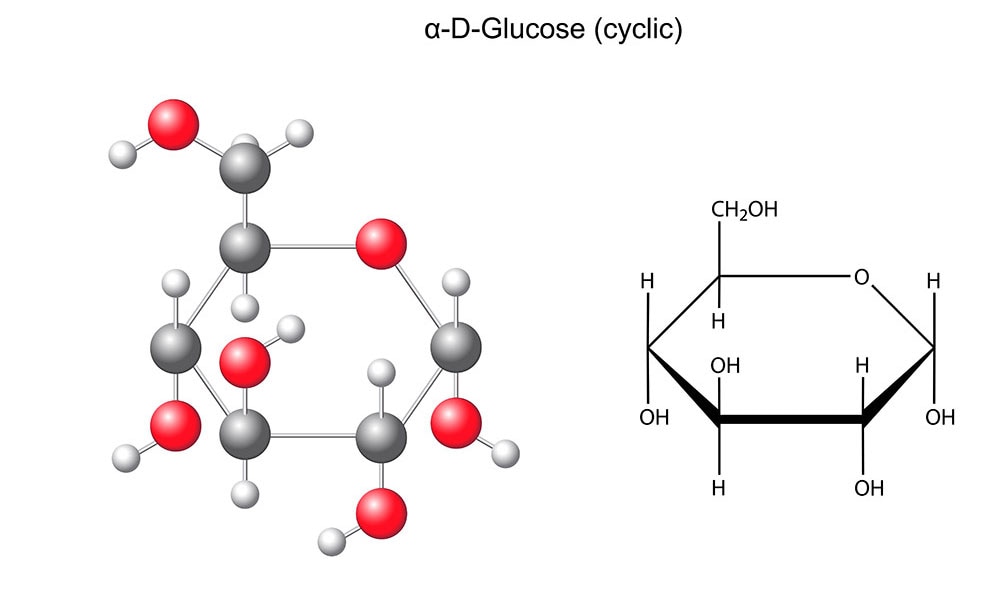
Glucose
Glucose is one of the fundamental molecules of life on Earth, the product of photosynthesis of solar energy stored in plants. Glucose is found either as a simple monosaccharide or as a constitutive unit of more complex molecules such as starch and the cellulose in fruit and vegetables, or glycogen in humans and animals. At the crossroads of energy metabolism, glucose can be considered to be the source of energy for our cells.
Source of energy and dietary fibre
Glucose belongs to the family of carbohydrates. It is a monosaccharide (simple sugar) naturally present in all living beings on Earth and is their most important source of energy. It is found in high quantities in fruit (including berries), vegetables and honey. When combined with other monosaccharides, such as fructose, it forms sucrose (table sugar) and lactose. Two glucose molecules form maltose, a disaccharide resulting from the hydrolysis of cereal starch. Maltose has slightly less sweetening power than sucrose. Athletes use it for a quick supply of energy, whereas in bakeries it is useful for the fermentation of leavened dough. Maltose is also found in the germinated cereal grains used to make many types of beer.
Starch consists of a large number of glucose molecules linked to each other in long chains. Cellulose is a polysaccharide made up of complex chains of starch. Unlike herbivorous mammals, the human body is unable to digest cellulose, so it serves as roughage in our diet.
Transforming solar energy
Glucose, a basic organic molecule, is synthesised by living beings capable of gluconeogenesis (animals) or photosynthesis (green plants, algae, some species of bacteria). Photosynthesis is a complex enzyme reaction making use of energy from sunlight, carbon dioxide (CO2) and water (H2O, sulphur bacteria use hydrogen sulphide instead of water) to produce glucose. It also releases oxygen, essential for life on our planet. Organisms capable of photosynthesis thereby make organic matter from inorganic matter. The resultant glucose can be found in its most simple form in fruit, as disaccharides in sugar beet or become the constitutive unit of starch or cellulose. This process transforms solar energy into food energy.
Syrup and solution
Glucose syrup is a thick, colourless syrup made by the hydrolysis of cornflour or potato starch. Confectioners use it in particular to make sweets and caramel, or in recipes in which the sugar must not be allowed to crystallise.
In medicine, a glucose solution is often administered intravenously to deliver energy quickly to a patient.
Nutrition
Like other carbohydrates, whether simple or complex, glucose has an energy value of 4 kcal per gram.
Starch is the main source of energy from our food. We start digesting it in our mouths. Salivary amylase, an enzyme found in saliva, starts the hydrolysis of starch and produces a mix of polysaccharides, maltose and glucose. This is the reason why, after a few seconds of chewing, a piece of relatively tasteless bread develops a certain sweet taste. This hydrolysis by amylase continues all the way to the stomach, then the maltase present in the small intestine splits the maltose molecules into two glucose molecules and completes the hydrolysis. The glucose is then actively absorbed by the cells of the intestine and enters the bloodstream. When it arrives in the liver, part of it is used to synthesise glycogen, an energy reserve in the form of a polysaccharide made up of tens of thousands of glucose units. This glycogen can then be converted back into glucose released into the bloodstream as and when the body needs it. This mechanism determines the level of glucose in the blood and is carefully controlled by two hormones, insulin and glucagon. These hormones inform the cells if they need to store or release glucose into the bloodstream to enable the body to function properly. The concentration of glucose in the blood plasma is called glycaemia. In a healthy person, blood glucose concentration is between 0.7 g and 1 g of glucose per litre of plasma. In a person with diabetes, blood sugar regulation is disrupted, leading to significant chronic peaks of hyperglycaemia (too much glucose in the blood) which, sooner or later, may present a threat to the person’s health.
GARRET, Reginald H. GRISHAM, Charles M., 2000. Biochimie. Torino: De Boeck Université.
REECE, URRY, CAIN, WASSERMAN, MINORSKY, JACKSON, 2012. Biologie. Canada. Pearson
CHUIT-GARCIA,C.F., SCHWEIZER, T.F., WURSCH, P.1987. Glucides Vevey. Nestec.S.A. Centre de documentation, département affaires publiques