Microscopic guests
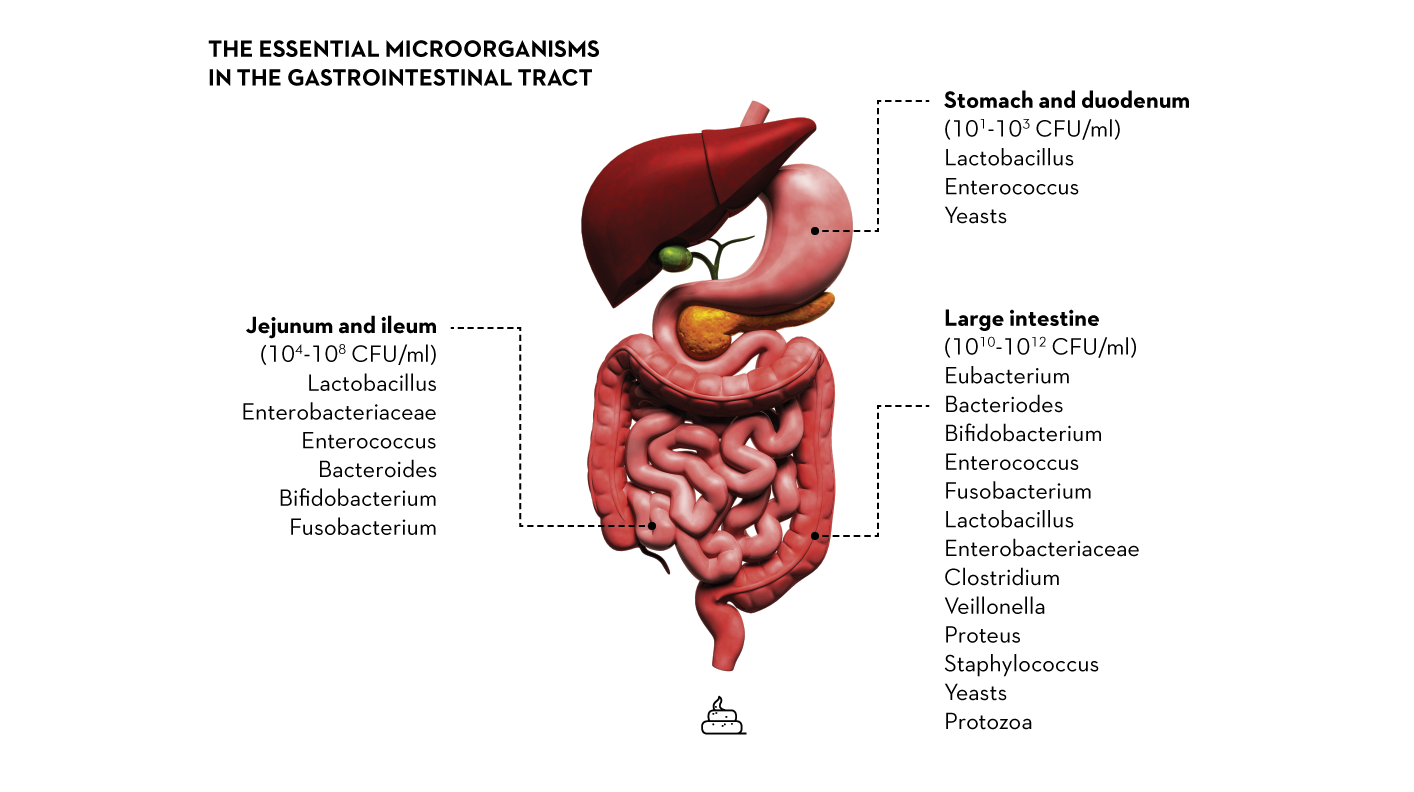
Our digestive system is like a biochemical factory. The pancreas sends enzymes to the mouth, the stomach and the gastrointestinal tract to break down the foods we eat and liberate their nutrients which will in turn feed our bodies and brains. Unused food residues leave our body via the colon as faeces.
I’m Microbiota, let me introduce myself
Within the gastrointestinal tract, the colon or gut is home to 100 trillion or so microbes, about 10 times more than all the cells in the human body. The average healthy adult has up to 2kg of bacteria in their gut, along with smaller numbers of microbes on the skin and all other parts of the body, together, these are ‘The Human Microbiota’.
The microbiota helps us digest foods that cannot be digested by the stomach and small intestine, and helps produce vitamins, such as B and K. A healthy and balanced microbiota will ensure our digestive systems are working properly. It also plays an important role in our immune system - an estimated 80% of our immune system can be found in the gut - by acting as a barrier as well as preventing aggression from other microorganisms(1).
Since the 1950s, microbiologists have shown increasing links between the gut microbiota, health and disease. The gut has been identified as a very important bioreactor for our health. The microbiota also has a permanent two-way message exchange with our brain on how we feel, on our digestion, metabolism, memory, mood, and wellbeing.
When we are born, our gastrointestinal tract is almost sterile. Microbes start to colonise our gut within the first minutes after birth. Understanding the first microbes that settle inside our gut is a crucial piece of the puzzle. One defining factor is the way we are born. Vaginal delivery pushes a baby through the lower part of its mother’s body, where it collects various microbes. Then, placed on its mother, the baby picks up more microbes from her skin. Babies born by caesarean will gather microbes from contact with the mother especially if breast-fed and will also collect them from the surroundings.
At birth, our immune system and gut ecosystem are still immature and need to learn to recognise the difference between harmful compounds (e.g. pathogens) and harmless ones (e.g. food proteins), reacting to the harmful ones and protecting us from disease while ignoring the harmless ones. The microbiota plays a role in helping our immune system mature and react appropriately to pathogens. One way to illustrate the importance of this role at birth is to compare babies delivered vaginally and those delivered by c-section. Their microbiota develops differently, so scientists can assume that the immune systems of each also develop differently. Studies show that babies born via caesarean may be more susceptible to certain infections and allergies early in life(2).
Many factors can change the microbiota's composition. Lifestyle changes play an important role, with elements such as urban life, sedentary habits, alcohol, smoking, drugs, as well as what we eat or don't eat can determining how the microbiota evolves(3).
Scientists even question whether the fact that modern life has helped us be "ever-cleaner and healthier" is a good thing. They suggest that increased hygiene levels may interfere with our microbiota. Antibiotics may be essential to fight infections, but we now know they upset the gut microbial balance. The microbiota was producing its own antibiotics centuries before Penicillin was discovered in 1928!
To simplify an extremely complicated matter, scientists group humans into three main enterotypes (similar to blood types) based on their microbiota: Prevotella, Bacteroides and Ruminococcus. The enterotypes are named after the most prevalent type of bacteria in each. Though enterotypes aren't explained by body mass, gender, age or even nationality, there may be a link with long-term diet. People who mostly eat carbohydrates have mainly Prevotellas and those who eat mostly protein and animal fats have mainly Bacteroides(4).
A short-term diet change will not affect someone's enterotype. However, scientists are researching whether a significant long-term dietary change could lead to a shift from one enterotype towards another and how this could make someone healthier.
Knowing your enterotype might one day be useful to find out which diet best suits you and even which drugs are the most effective!
Scientists are also wondering whether one's microbiota can be linked to a propensity to gain weight. A growing scientific opinion believes that it will soon be possible to fight obesity by modifying the microbiota.
One study conducted on mice gave encouraging results. Pairs of twin-sister mice, one thin and the other one obese, were used as microbiota donors to germ-free mice. The germ-free mice receiving microbiota from obese sisters quickly gained weight with less food, while those getting microbes from lean sisters stayed thin. The difference on metabolism was significant(5).
Ridaura’s mouse model also shows that obese mice, with microbiota transplants and a tailored diet, can lose weight. A recent review strongly supports that the findings in this study are a step closer to testing anti-obesity therapeutics for humans using specific combinations of the right bacteria(6).
Better understanding our microbiota, the different strains of bacteria that have taken up residence in our guts and the role they each play on how food is processed in our bodies will definitely influence the way we eat in the future.